DSS: Redefining Biotechnology & Life Science in India
- About Us
- Products & Services
PRODUCTS & SERVICES
- Applications & Specialities
All Applications & Specialities
- Brands
- Contact Us
-
-
0
- ☰
- About Us
- Products & Services
-
Kits Reagents & Consumables
- Cytogenetics
- Dyes
- Fluorescence In Situ Hybridization (FISH)
- High-Performance Liquid Chromatography (HPLC)
- Histology
- Immuno Histo Chemistry (IHC)
- IVF Consumables
- Molecular Pathology & Diagnostics
- Multiplex Ligation-Dependent Probe Amplification (MLPA)
- Nucleic Acid Extraction
- PharmDx
- Real Time PCR
- Special Stains
- Instruments
- Software
- Accessories
- Advanced Material
- Therapies
-
Kits Reagents & Consumables
- Applications & Specialities
- Brands
- Brand - Life Sciences
- 3i
- ABBERIOR INSTRUMENTS
- Abbott Molecular
- ADS Biotec
- APPLIED SPECTRAL IMAGING
- BioAir Tecnilabo
- DAKO (AGILENT)
- Eden Tech
- Elveflow
- ENTROGEN
- EUROCLONE
- EVIDENT
- Genea
- Hamamatsu Photonics
- Invivoscribe
- MASTER DIAGNOSTICA
- MBF BIOSCIENCE
- Medical Tek Co. Ltd
- MILESTONE MED SRL
- Molecular Machines & Industries
- MRC HOLLAND
- NeoDx
- Onward Assist
- Profound
- SCIENTIFICA
- SpaceGen
- Seqlo
- µCyte
- Brand - Industrial
- Brand - Life Sciences
- News & Events
- Career
- Contact Us
- Testimonial
- Blogs
- R&D
- CSR
- Press Release

World Health Day 2026: Strengthening Universal Health Coverage Through Science and Diagnostics
BY Ms. Janhavi Srirangaraj, Product Specialist 7th April 2026
Every year on April 7, the global community pauses to observe World Health Day, marking the founding of the 1st World Health Assembly, the World Health Organization (WHO) in 1948. This day not only honours the birth of the world’s apex health governance body but also spotlights a critical global health challenge chosen by WHO. In 2026, the focus is on “Global Action for Universal Health Coverage”, a theme that aligns closely with WHO’s year long campaign motto: “Together for health. Stand with science.”
This theme underscores an urgent idea: everyone, everywhere, should have access to quality, evidence based healthcare, right from prevention and diagnosis to treatment and rehabilitation, without facing financial hardship. Universal Health Coverage (UHC) is more than a policy goal; it is a commitment to equity, resilience, and trust in science driven health systems.
Why World Health Day Matters in 2026
World Health Day 2026 is being spearheaded by WHO to galvanise policymakers, researchers, healthcare providers, diagnostic companies, and communities around the goal of accessible, affordable, and timely healthcare for all. The theme “Global Action for Universal Health Coverage” invites governments to design financing, governance, and service delivery models that leave no one behind, especially marginalised and rural populations.
Diagnostics: The Backbone of Universal Health Coverage
Underpinning UHC is diagnostics, the science that turns suspicion into evidence, uncertainty into action. Modern diagnostic tools, from rapid lateral flow tests to molecular platforms such as PCR and next generation sequencing (NGS), enable early detection, accurate outbreak surveillance, and personalised care across the One Health continuum. Molecular diagnostics, in particular, generate the data that inform vaccines, treatments, and public health policies. By identifying antimicrobial resistant pathogens or climate linked infectious agents, diagnostics rebuild trust in science and support laboratories worldwide—from global hubs to resource limited Indian labs working in oncology and infectious disease testing.
In this context, India’s diagnostic ecosystem has emerged as a compelling success story. During the pandemic, the country rapidly scaled up RT PCR testing and now continues to innovate with home grown solutions, global partnerships, and point of care technologies that extend precision diagnostics beyond metro centres into district labs and rural clinics.
DSS Imagetech: Where WHO’s Theme Comes Alive
As a trusted leader in molecular diagnostics and life sciences solutions with over two decades of innovation, DSS Imagetech embodies the mission of “Together for health. Standing with science”, we strive to make a meaningful difference across infectious diseases, oncology, genetic disorders, and reproductive health. As a molecular diagnostics and life sciences solutions company, DSS Imagetech translates advanced science into practical tools that support universal access to accurate, timely diagnostics, which is a cornerstone of UHC.
Our Clinical Diagnostics Division, established in 2003, offers solutions for cancer diagnosis, prenatal and postnatal genetic aberration testing, and infectious disease workups (HIV, HBV, HCV, CMV, sepsis, and more). Technologies such as FISH (Fluorescence In Situ Hybridisation), immunohistochemistry (IHC), Real Time PCR for viral load, Integrated Chromosome Profiling, MLPA, and karyotyping are deployed to detect, characterise, and monitor disease at the molecular level.
For WHO’s 2026 theme, this means ensuring that the scientific tools needed for diagnosis and treatment decisions are not confined to elite centres, but are progressively available, validated, and accessible to clinicians and patients across India and beyond.
Infectious Disease Diagnostics: From Labs to Communities
At DSS Imagetech, infectious disease diagnostics sit at the heart of our mission to strengthen public health and support universal health coverage. By combining high‑sensitivity molecular tools, streamlined workflows, and scalable detection platforms, we help clinicians and laboratories detect, monitor, and contain infections earlier and more accurately—wherever care is delivered.
Abbott Molecular Real‑Time PCR Assays
DSS Imagetech distributes Abbott Molecular Real‑Time PCR assays for HCV, HBV, HIV, EBV, and CT/NG across India, turning molecular diagnostics into a frontline defence against chronic and sexually transmitted infections. For these diseases, the difference between a molecular diagnosis and a missed diagnosis can mean years of preventable suffering, transmission, or long‑term complications. The Abbott RealTime High Risk HPV assay supports evidence‑based cervical‑cancer screening, integrating early‑stage infection detection into routine women’s health programmes. The HIV‑1 Qualitative assay, using dried blood spot sampling, extends reliable infant diagnosis beyond well‑equipped urban labs into remote and resource‑limited settings, where early treatment is critical for survival and prevention of vertical transmission.
Together, these tools actively support India’s drive toward universal screening and early treatment, reducing the burden of chronic viral infections and strengthening public‑health surveillance systems that are central to both Universal Health Coverage (UHC) and the One Health approach.
VIASURE RT‑PCR: High‑Throughput Molecular Surveillance
Complementing targeted assays, CerTest’s VIASURE RT‑PCR platform offers a complete end‑to‑end solution—from RNA/DNA extraction through automated PCR setup to integrated result interpretation—producing over 200,000 diagnostic tests per day. Its broad menu covers gastrointestinal and respiratory infections, antimicrobial resistance, tropical and vector‑borne diseases (including dengue, chikungunya, typhoid, and TB), sexual‑health infections, and meningitis, directly addressing the high‑burden conditions that shape India’s public‑health landscape.
By simplifying workflows and enabling high‑throughput testing, VIASURE empowers laboratories of all sizes—from small district labs to large reference centres—to deliver molecular‑level answers at scale. This capacity is a critical enabler of UHC and One Health surveillance, allowing faster outbreak detection, more accurate antimicrobial‑resistance monitoring, and more responsive clinical decision‑making in resource‑diverse settings.
NeoDx Infectious Disease PCR Kits
Under the broader infectious‑disease diagnostics umbrella, NeoDx’s infectious disease PCR kits, available through DSS Imagetech, further expand the toolbox for targeted molecular testing. These CE‑IVD‑certified kits are designed for a range of viral and bacterial infections, supporting rapid, accurate detection in both routine and outbreak‑related scenarios. By integrating NeoDx’s PCR solutions into the existing portfolio of Abbott and CerTest platforms, DSS Imagetech reinforces a multi‑layered, evidence‑based approach to infectious‑disease management—linking point‑of‑care‑ready assays with high‑throughput molecular workflows to serve everything from individual patients to population‑level surveillance programmes.
In this way, infectious disease diagnostics at DSS Imagetech are not just about technology; they are about protecting communities, preventing transmission, and enabling science‑driven, equitable healthcare access—a vision that aligns fully with WHO’s 2026 theme of “Together for health. Stand with science.”
Cytogenetics Diagnostic Solutions: From Probes to Automated Workflows
DSS Imagetech provides a complete cytogenetics ecosystem—spanning FISH probes, culture media, automation instruments, karyotyping software, and imaging systems—designed to streamline workflows, enhance accuracy, and support both prenatal and postnatal genetic analysis. This integrated portfolio ensures laboratories can deliver rapid, reliable chromosome analysis for conditions ranging from congenital disorders to haematological malignancies, with automation reducing hands-on time and improving reproducibility.
Abbott Vysis FISH Probes & IntelliFISH
Our cytogenetics solutions feature Abbott Vysis DNA FISH probes, available in India through DSS Imagetech, covering haematology, oncology, and prenatal applications. Key probes include the AneuVysion DNA Probe Kit for rapid prenatal aneuploidy detection (trisomies 13, 18, 21, X, Y), PathVysion HER‑2 DNA Probe Kit for breast cancer HER2 amplification, Vysis ALK Break Apart FISH Probe Kit for lung cancer, Vysis CLL FISH Probe Kit for chronic lymphocytic leukaemia, LSI BCR/ABL Dual Color Dual Fusion, PML/RARA, and more. With over 300 probes tailored for solid tumours (breast, lung, bladder) and haematological malignancies, plus ancillary reagents like paraffin pretreatment kits, VP 2000 reagents, and DAPI counterstains, these tools enable precise detection of deletions, amplifications, and translocations at every stage of the patient journey.
CyFISH by NeoDx
CyFISH, NeoDx’s proprietary FISH probe portfolio distributed by DSS Imagetech, brings Indian‑developed, internationally benchmarked probes for haematology, solid tumours, and prenatal screening (Indian IVD registration in progress). Highlights include HER‑2 amplification (NCHHG22001) for breast cancer, ALK fusion (NCHAG22002) for lung cancer/lymphomas, BCR/ABL (NCBAG22003), PML/RARA (NCPRG22005), AML1/ETO (NCAEG22004), ETV6/RUNX1 (NCERG22006), CBFB break‑apart (NCCBG22020), IGH break‑apart (NCIGH22019) for lymphoma, 1p/19q deletion (NC1PQ22007) for oligodendroglioma, and prenatal chromosomes probe (NCPCP22008) for aneuploidies in high‑risk pregnancies. RUO probes perform on FFPE, bone marrow, and amniotic fluid samples, reducing import dependency while upholding global standards.
EuroClone Culture Media and Reagents
For robust cell culture, EuroClone supports postnatal (Chromosome Kit P/M, ChromoLymphoB, EKAMTB100/500 media) and prenatal (Amniopan, AmnioFlask, Amnioslide) workflows, yielding up to 100 tests/bottle. Includes antibiotics, L‑Glutamine, Colcemid, HEPES, trypsin, and disposables (Primo flasks/dishes/tubes) for high‑quality metaphase spreads compatible with karyotyping and FISH.
Automation for FISH and Hybridization
Abbott ThermoBrite (12 slides, 30–99°C programmable, rapid ramping <3 min to 95°C, humidity control) and EuroClone HyperChrome (denaturation/hybridization modes, ±1°C accuracy, 12 slides, touch‑screen) automate FISH processing, cutting hands-on time >50%, ensuring uniform temperature/humidity, and supporting co‑denaturation for low‑background signals. Hanabi-S1020 Auto Chromosome Staining processes 20‑slide batches continuously for G‑banding efficiency.
Advanced Imaging and Karyotyping Software
Applied Spectral Imaging (ASI) software like GenASIs HiSKY enables hyperspectral karyotyping with unique chromosome colouring, while CytoPower automates digital karyotyping, FISH analysis, and scanning. Automatic Metaphase Finder scans slides, captures optimal metaphases, and boosts throughput—transforming manual processes into reproducible, high‑volume workflows.
This end‑to‑end cytogenetics portfolio—from culture to automated analysis—empowers Indian labs with speed, precision, and scalability, advancing prenatal screening, cancer diagnostics, and hereditary disorder management in alignment with WHO’s science‑led health equity goals.
Oncology Diagnostic Solutions: qPCR and NGS from DSS Imagetech
DSS Imagetech delivers precision oncology through targeted qPCR and comprehensive NGS solutions from leading partners EntroGen, NeoDx, and SpaceGen, enabling clinicians to detect mutations, fusions, and genomic alterations that guide personalised treatment decisions. These molecular tools—from rapid mutation panels to deep tumour profiling—bridge routine diagnostics with advanced therapy selection, supporting India’s growing demand for evidence-based cancer care.
EntroGen: Targeted qPCR Mutation Panels
EntroGen’s CE-IVD certified qPCR kits, distributed exclusively by DSS Imagetech, provide fast, reliable mutation analysis for major cancers including colorectal (KRAS, NRAS, BRAF, PIK3CA, RAS), lung (EGFR, PIK3CA, cell-free EGFR), breast (PIK3CA, BRCA Complete Expanded Panel), leukaemia (BCR-ABL P210/P190, AML1-ETO, PML-RARA, TEL-AML1, CBFb-MYH11, MLL-AF4), melanoma (BRAF, NRAS, c-Kit), thyroid, glioblastoma (MGMT methylation, IDH1/2, EGFR V3), and GIST. These single-round PCR assays deliver results in under 2 hours on standard qPCR platforms, making them ideal for smaller labs while supporting companion diagnostic decisions for targeted therapies like EGFR inhibitors or BRAF blockers.
NeoDx: Complementary qPCR and Molecular Tools
Through NeoDx’s infectious disease PCR kits and oncology-focused molecular assays available via DSS Imagetech, labs gain additional qPCR flexibility for rapid pathogen detection alongside cancer biomarker screening. While primarily known for CyFISH probes, NeoDx’s PCR portfolio extends to haematological and solid tumour applications, integrating seamlessly with EntroGen panels for labs handling both infectious complications and cancer molecular profiling.
SpaceGen: Comprehensive NGS Genomic Profiling
SpaceGen’s NGS panels represent the pinnacle of tumour profiling, analysing 1,200 genes in the Tumor Comprehensive Genomic Profiling Panel to detect SNVs, indels, copy number variations (CNVs), and gene fusions across solid and haematological malignancies. The Hereditary Cancers Panel screens 58 clinically actionable genes, while the BRCA1/2 Gene Mutations Detection Kit identifies hereditary risks for breast, ovarian, and pancreatic cancers—empowering preventive strategies. These panels support liquid biopsy workflows with cell-free DNA isolation kits, enabling non-invasive monitoring of treatment response and minimal residual disease (MRD).
Integrated Workflow: From qPCR Screening to NGS Confirmation
Together, EntroGen’s rapid qPCR panels provide cost-effective initial screening, NeoDx PCR adds specialised applications, and SpaceGen NGS delivers gold-standard genomic insights—forming a tiered oncology strategy. This progression allows district hospitals to start with targeted mutation tests, while tertiary centres perform full profiling, ensuring actionable results at every resource level. By making precision diagnostics accessible beyond metro cities, DSS Imagetech advances WHO’s vision of science-led universal health coverage, where every cancer patient benefits from molecular evidence tailored to their tumour’s unique biology.
Histopathology Diagnostic Solutions: From Tissue to Diagnosis
DSS Imagetech delivers a complete histopathology continuum—from tissue collection through processing, staining, companion diagnostics, and digital imaging—powered by Milestone Medical, Agilent Dako, and Evident (Olympus). This integrated ecosystem ensures every specimen maintains optimal quality for accurate oncology diagnosis, IHC, FISH, and NGS, supporting faster turnaround times and evidence-based treatment decisions across Indian pathology labs.
Milestone Medical: Pre-Analytical Excellence
Milestone Medical, distributed exclusively by DSS Imagetech, pioneered microwave tissue processing and offers tools covering fixation, decalcification, grossing, sectioning, and frozen sections. The LOGOS EVO dual-retort processor uses xylene-free, microwave-hybrid technology for rapid, validated protocols that preserve morphology and molecular integrity—processing urgent specimens multiple times daily while cutting reagent costs and enhancing safety. Complementary systems like FixSTATION (controlled formalin fixation), BoneSTATION (bone decalcification for NGS-ready biopsies), HistoDream microtomes, and PrestoCHILL/FineFIX (frozen sections in under 10 minutes) create a lean, scientifically validated pre-analytical chain that eliminates bottlenecks and protects downstream results.
Agilent Dako: IHC Standards and Companion Diagnostics
Since 2007, DSS Imagetech has brought Agilent Dako’s global IHC leadership to India, featuring the antibodies, Autostainer Link 48 (48 slides in <3 hours, up to 96/day) for automated, high-throughput staining with FLEX antibodies and EnVision FLEX visualisation. The Dako CoverStainer fully automates H&E processing—from dewaxing to coverslipping—for consistent routine diagnostics.
Key companion diagnostics include PD-L1 IHC 22C3 pharmDx (Tumour Proportion Score for pembrolizumab eligibility in NSCLC). With extensive primary antibodies, EnVision DuoFLEX doublestain, and ancillaries, Dako enables precise prognostic, proliferation, and tissue characterisation testing that meets international accreditation standards.
Vitro Antibodies: Specialised IHC Reagents
Vitro Antibodies, distributed by DSS Imagetech, expands IHC capabilities with a curated selection of high-specificity primary antibodies for tumour markers, prognostic indicators, and tissue identification. Optimised for automated platforms like Autostainer Link 48, these reagents support routine diagnostics and research applications in oncology, providing cost-effective alternatives that maintain validated performance for Indian pathology labs handling diverse caseloads.
Evident (Olympus): Precision Imaging Infrastructure
Evident (formerly Olympus) microscopes—upright, inverted, and fluorescence models—provide the optical foundation for H&E review, cytology, FISH analysis, and research confocal imaging, trusted by eminent Indian institutions for decades of reliable performance & support.
The SLIDEVIEW VS200 whole slide scanner digitises slides in brightfield, fluorescence, polarisation, darkfield, and phase contrast modes (up to 100x magnification, True Color LED, X Line objectives), supporting DICOM-LIMS integration, TruAI deep-learning analysis, remote consultations, and tumour board reviews. Field-upgradeable for fluorescence or high-capacity loaders, it brings digital pathology to Indian labs, enabling quantitative analysis, annotation, and secure archiving.
Together, Milestone’s pre-analytics, Dako’s staining expertise, and Evident’s imaging form an end-to-end histopathology solution—from biopsy to digital diagnosis—that ensures every tissue specimen yields maximum diagnostic value. This workflow empowers pathologists with precision, consistency, and scalability, directly advancing WHO’s call for science-led universal health coverage in cancer care.
MLPA Overview: Detecting Genomic Disorders Through Copy Number Analysis
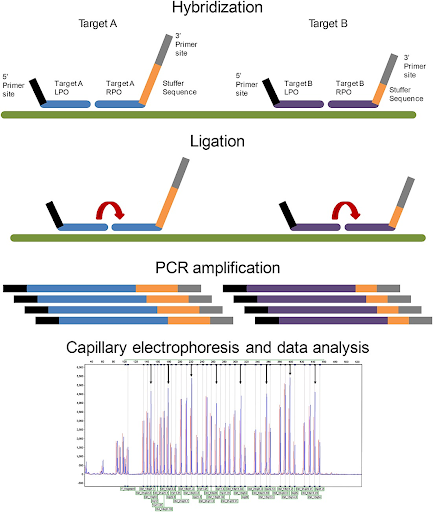
MLPA (Multiplex Ligation-dependent Probe Amplification) from MRC Holland, distributed by DSS Imagetech, is a multiplex PCR technique that detects copy number variations (CNVs)—deletions or duplications across up to 50 genomic targets—in a single reaction using just 20 ng of DNA, making it ideal for diagnosing genomic disorders invisible to karyotyping. MLPA uses locus-specific probes that hybridise adjacently, ligate only when perfectly matched, and amplify via universal primers for precise gene dosage quantification, excelling at microdeletions/duplications in conditions like Duchenne Muscular Dystrophy (65–70% of cases), Spinal Muscular Atrophy (SMN1), hereditary breast/ovarian cancer (BRCA1/2), Charcot-Marie-Tooth (PMP22), Prader-Willi/Angelman (imprinting), and microdeletion syndromes (DiGeorge/22q11, Williams/7q11). Compared to karyotyping, MLPA resolves submicroscopic changes (<5 Mb); versus NGS, it offers superior detection of large multi-exon CNVs in repetitive regions at 10x lower cost with 24-hour results via capillary electrophoresis, enabling high-throughput prenatal/postnatal testing that confirms array CGH findings, guides family counselling, and supports Universal Health Coverage through cost-effective, actionable genetic insights where dosage determines disease.
Standing with Science, Together for Health
On World Health Day 2026, DSS Imagetech reaffirms its commitment to “Global Action for Universal Health Coverage” and the broader WHO call to “Stand with science.” By combining cutting edge molecular diagnostics, home grown innovation, and scalable solutions for infectious disease control and precision oncology, we help ensure that evidence based, accurate diagnostics are not a privilege, but a right—accessible to clinicians and patients across the healthcare spectrum.
This is the promise of universal health coverage: no one left behind, no decision made in the dark, and every community empowered by science. On April 7 and every day thereafter, DSS Imagetech stands with labs, doctors, patients, and global partners—because truly healthy societies are built on trust in science, equity in access, and collaboration across borders. Explore our 2026 solutions for universal health coverage and join us in advancing evidence based diagnostics that reach every corner of healthcare.
References
The following references support the themes and information presented in the World Health Day 2026 blog post on strengthening universal health coverage through diagnostics and science.
- World Health Organization. (2025). World Health Day 2026: Global Action for Universal Health Coverage. WHO Press. (Outlines the 2026 theme “Together for health. Stand with science,” emphasizing diagnostics in equity and One Health.)
- World Health Organization. (2024). Universal Health Coverage (UHC) Factsheet. Available at: who.int/health-topics/universal-health-coverage. (Details UHC pillars: prevention, diagnosis, treatment without financial hardship, with India case studies on RT-PCR scaling.)
- Abbott Molecular. (2025). RealTime PCR Assays for HIV, HBV, HCV, HPV: Technical Manual. Abbott Diagnostics. (Covers qualitative HIV-1 DBS assays and High Risk HPV for cervical screening in resource-limited settings.)
- CerTest Biotec. (2024). VIASURE RT-PCR Kits: Validation for High-Throughput Infectious Disease Panels. CerTest Whitepaper. (Highlights GI/respiratory/TB/dengue menus for scalable surveillance in district labs.)
- Abbott Vysis. (2026). FISH Probes Catalog: AneuVysion, PathVysion HER-2, ALK Break-Apart. Abbott Oncology. (Over 300 probes for prenatal aneuploidy, solid tumors, hematology; includes IntelliFISH automation.)
- NeoDx Biotechnology. (2025). CyFISH Probe Portfolio: Indian RUO Probes for Oncology and Prenatal. NeoDx Datasheet. (Details NCHHG22001 HER-2, NCBAG22003 BCR/ABL; FFPE/bone marrow compatibility.)
- MRC Holland. (2024). MLPA Technology: Copy Number Detection for Genomic Disorders. MRC Holland Manual. (Probes for DMD/SMN1/BRCA; advantages over NGS for large CNVs at low cost.)
- EuroClone. (2025). Chromosome Culture Media: Amniopan, ChromoLymphoB Protocols. EuroClone Catalog. (Postnatal/prenatal media yielding 100 tests/bottle for metaphase spreads.)
- EntroGen Inc. (2026). qPCR Mutation Panels: KRAS/EGFR/BRCA for Precision Oncology. EntroGen CE-IVD Guide. (Single-round assays <2 hours for colorectal/lung/breast cancers.)
- Agilent Dako. (2025). Autostainer Link 48 and PD-L1 IHC 22C3 pharmDx. Dako Companion Diagnostics. (High-throughput IHC for NSCLC pembrolizumab eligibility.)
- Milestone Medical. (2024). LOGOS EVO Microwave Processor: Xylene-Free Tissue Prep. Milestone Protocols. (Preserves morphology for NGS/IHC/FISH in urgent biopsies.)
- Applied Spectral Imaging (ASI). (2025). GenASIs HiSKY and CytoPower: Hyperspectral Karyotyping. ASI Software Manual. (Automated metaphase finding and FISH analysis.)
About the Writer
Ms. Janhavi Srirangaraj, Assistant Manager – Product & Application,Integrated Diagnostic Division at DSS Imagetech. With an MSc in Agrobiotechnology from Justus-Liebig-University, Germany, and over six years of global experience in Switzerland and Germany, she brings expertise in molecular biology, assay development, and advanced diagnostics. Janhavi has significantly contributed to establishing SARS-CoV-2 testing labs in Switzerland and continues to drive innovation in molecular diagnostics, including MLPA technology.
Latest Articles
World Health Day 2026: Strengthening Universal Health Coverage Through Science...
BY Ms. Janhavi Srirangaraj, Product Specialist 7th April 2026
Every year on April 7, the global community pauses to observe World Health Day, marking the founding of the 1st World Health Assembly, the World Health Organization (WHO) in 1948....
Read MoreWorld Tuberculosis Day: “Spreading Awareness, Saving Lives”
BY Mr. Satendra Saxena, Application Specialist, DSS Imagetech 23rd March 2026
Every time on March 24, the world pauses to observe World Tuberculosis (TB) Day. It’s a day of mixed feelings. We celebrate the scientific improvements that have made this complaint...
Read MoreInternational Women’s Day – Prioritising Women’s Health for a Healthier...
BY Ms. Sohini Chatterjee, Application Specialist, DSS Imagetech 7th March 2026
In 2013, one public disclosure transformed the global conversation around women’s health. When Angelina Jolie revealed in a New York Times op-ed that she carried a BRCA1 mutation and had...
Read More